ADOLFO SARTINI WASN’T SUPPOSED TO DIE from the flu. He was 29 years old, healthy and strong; he had answered the draft in 1917 and was picked to work as an Army engineer. And yet, the things that should have protected Sartini actually made him an utterly typical victim of the virus that killed him: the notorious 1918 “Spanish” flu.

Adolfo Sartini in uniform
Extending over the world in three deadly waves, the 1918 flu infected some 500 million people and killed at least 50 million—as much as 5 percent of the world’s population, though it is hard to pin down a precise figure—placing it among the deadliest pandemics ever recorded. So many died so quickly that cities ran out of coffins. Extra gravediggers were called up by the hundreds, and when there weren’t enough gravediggers, steam shovels came in to dig mass graves for the bodies that were piling up in morgues and on roadsides.
The virus piggybacked on World War I troop movements and was sustained by soldiers’ close quarters, yet it also spread to people and places far removed from the war. And the world was all but defenseless against it: without vaccines, antibiotics or antivirals, patients made do with rest, fluids, fresh air and prayer.
The 1918 flu was a killer, and it was also a puzzle. Unlike most flu strains, which are deadly mostly to the very young and the very old, the 1918 flu disproportionately struck down young adults. Graph typical seasonal flu deaths according to age, and you get a “U” shape, with high mortality among babies on the left and the elderly on the right, and a strong, healthy trough in the middle. But when you plot out deaths from the 1918 flu, you get a “W” instead, with the peak centered near age 28—almost exactly Adolfo’s age when he died.
For decades, researchers have been trying to understand what made young adults like Sartini so vulnerable to this particular flu. Maybe people over 40 had already been exposed to similar strains and built up some immunity, while younger ones were immunologically unprepared. Maybe soldiers like Sartini were already weak from other infections, like tuberculosis. Maybe young, healthy people suffered from an overactive immune response—a so-called cytokine storm, named for proteins that help direct the body’s immune response—that damaged their lung tissue and allowed fatal infections to establish themselves. Or maybe it was all of those things at once.

Rows of tents at Emery Hill in Lawrence, Massachusetts, where victims of the 1918 influenza pandemic were treated. —Photo courtesy of the National Archives
It’s a puzzle that fascinates Ruth Craig ’74. That’s because Sartini’s story embodies her two life’s passions: molecular biology, which she researched and taught for more than 20 years from her labs at Johns Hopkins University School of Medicine and the Geisel School of Medicine at Dartmouth, and genealogy, which she discovered later on and took up as a second career when she moved from active to emeritus professorship. “The two tracks seem very different but actually intersect,” says Craig, “and that intersection is the flu.”
IN 1987, AS A BRAND NEW FACULTY MEMBER at Johns Hopkins University School of Medicine, Craig led a research team that discovered a gene that helps control whether cells live or die. The gene, called myeloid cell leukemia-1, or MCL1, is a member of a family of genes with similar jobs, and their involvement in leukemia surprised cancer researchers—it suggested that cancer isn’t just about runaway cell growth, but also about cells that fail to die when their time is up.
But MCL1’s influence goes beyond cancer, as Craig discovered over the course of many years examining the gene. In 2005, Craig was part of a team led by David Dockrell, an infectious disease researcher at the University of Sheffield, that looked at how MCL1 helps immune cells fight infection. They found that MCL1 makes a protein that helps signal immune cells called macrophages to attack bacteria. The macrophages swallow up the invaders, and they also recruit other immune cells to join their offensive. When levels of the MCL1 protein drop, macrophages get the signal to stand down and die off, taking their infectious “prisoners” with them and calling off the immune attack. The response is exquisitely tunable and can be turned on and off in different kinds of cells with laser precision and speed. But what would happen if, for some reason, it failed to turn off on time?
Craig and Dockrell tried to answer that question. Their team started with mice with an MCL1 gene that caused them to overproduce the MCL1 protein. Then, the researchers gave those mice bacterial lung infections. Mice with the modified gene could not clear the infection. Moreover, their lungs were overwhelmed with inflammatory cells. The macrophages, it seemed, didn’t know when to die—so while the immune system kept up its attack, the bacteria went on multiplying, and the mice got sicker and sicker.

Ruth Craig ’74 and Bob Sartini visit St. Michael Cemetery in Boston, where Spanish flu victim Adolfo Sartini is buried.
This is where Craig’s gene intersects with Sartini’s story. That’s because if he was like most flu victims, Sartini didn’t actually die from the flu, but from a bacterial lung infection that set in afterward. Craig wondered: Could he have died—too young, too early—in part because some cells in his body died too late?
BOB SARTINI KEEPS HIS GREAT-UNCLE Adolfo’s Army chest in his living room in Vermont; on the wall of his Boston apartment, he hung oval-framed pictures of his grandparents and a memorial certificate from the U.S. Army commemorating Adolfo’s supreme sacrifice in World War I. When his grandfather died, Bob says, the certificate “was basically on the trash heap,” Bob became friends with Craig in the late 1970s, when they worked down the hall from each other at Boston University School of Medicine (Bob, who is now retired, spent his career there), and she had the certificate framed for him as a birthday present. “From then on, it’s been on the wall in my house.”
Bob imagined filling up Adolfo’s old Army chest with “period things,” artifacts that would tell the story of Adolfo’s life, but the family lore was meager. Bob knew Adolfo had followed his brother Eugenio, Bob’s grandfather, from Italy to America. He knew that he had spent time working at a country club in Newton, Massachusetts, before enlisting in the Army, and he knew that Adolfo had died young, of the flu. But he wondered: Why did Adolfo enlist? Did Adolfo know that because he was not a U.S. citizen and had not w declared an intention to become one, he was not required to answer the draft? Did he deliberately pass up this exemption so that he could step forward to defend his chosen home? And how to make sense of the irony of a healthy soldier being struck down not by shells or machine guns but by something as mundane and typically benign as the flu?

Ruth Craig ’74 places flowers on the grave of Adolfo Sartini, who died of the Spanish flu in 1918.
Bob wondered about this off and on over the years, going so far as to contact the National Military Personnel Records Center, but they could offer no help: Adolfo’s records had burned up in a 1973 fire that destroyed more than 16 million Army and Air Force personnel files.
Then, around 2013, Craig mentioned that she was working on becoming a certified genealogist and needed a project—something specific and preferably something in Massachusetts, where, unlike New Hampshire, birth and death records weren’t kept sealed. Adolfo seemed perfect.
Craig began by tracking down the register for the ship that brought Adolfo to America. The register listed his birthplace in a farming region of Italy. Then, from her desk at Dartmouth, she pored over digitized images of 19th-century Italian record books. Finally, she found Adolfo’s birth record and birthdate: Feb. 8, 1889.
But what Craig really wanted to find was his death certificate. Not sure where to look next, she posted to an online military history forum, where an expert in World War I history pointed her to a military base that trained engineers, Camp A.A. Humphreys, in Virginia. A search of Virginia death records confirmed it: Adolfo died at the Virginia training camp, far from home and far from the front lines, of a sickness that was one thing that the broken world had in common.
While Craig was searching genealogical records, she was also poring over the scientific literature on the flu, hoping to answer the question that was rising up in her mind: Why did it kill someone like Adolfo?

Soldiers parade in front of the Carnegie Building on Armistice Day, Nov. 11, 1918, wearing surgical masks to protect themselves from the Spanish flu.
The Spanish Flu at Pomona
It was Nov. 11, 1918—Armistice Day. The global catastrophe that was World War I was finally over, and people everywhere were celebrating. But as soldiers paraded down College Avenue in Claremont, it wasn’t hard to see that the celebration was tempered by worry and caution. Every man in the parade was wearing a white surgical mask to protect himself from the scourge that was striking down the young and fit across the country.
But whether through luck or caution, the pandemic claimed only one life on the Pomona campus. A previously healthy young woman named Viola Minor Westergaard, the wife of Pomona faculty member Waldemar Westergaard, succumbed during the final throes of the epidemic, on Jan. 7, 1919. Viola’s parents later donated a collection of books and other items to Honnold-Mudd Library in her honor, including a bust of her face by artist Burt Johnson.
—Mark Wood

Bust of Viola Westergaard, the only victim of the Spanish flu on the Pomona campus
Flu viruses are always changing, accumulating small genetic mutations and, once in a while, making more radical shifts that constitute entirely new flu subtypes—not just variations on a theme but fresh, unfamiliar melodies. This year-to-year change explains why getting the flu this year doesn’t mean you’ll be protected against it next year. It also helps explain why older people are sometimes spared the worst of a flu that seriously sickens younger ones: Their immune systems remember similar strains that circulated decades ago and can mount some defense, even if it is an imperfect one.
For instance, some 80 percent of those who died from the 2009 “swine flu” pandemic were under 65—turning the expected mortality statistics for seasonal flus on their head. Why were younger people w likelier to get sick and die of swine flu? Researchers think that older people had been exposed to similar flus, including the 1918 flu, in the early 20th century, and therefore had some protection against the 2009 version.
Yet that can’t completely account for the 1918 flu’s W-shaped curve and the peculiar vulnerability of those who were born around 1889, like Adolfo. Craig’s search brought her to mathematician David Earn, who studies mathematical biology at McMaster University in Canada. Earn and his colleagues have explored the possibility that a person’s very first flu—the one he or she encounters as a baby—makes a more powerful impression on the immune system than any other. This hypothesis, called “antigenic imprinting,” goes back to the 1950s and offers an appealingly parsimonious explanation for the W-shaped curve. If it is correct, Earn wrote, it means that your risk of dying from the flu has everything to do with the biological “distance” between your first flu and the one you happen to be sick with right now.
IN THE FALL OF 1889, a new flu broke out in St. Petersburg, Russia. Quickly, the flu spread west. (It may have actually already made its way through India and Central Asia before being reported in Russia.) Though it wasn’t as deadly as the 1918 flu, the flu of 1889 and 1890 is recognized as the first pandemic of the connected world. Extensive railroads linked the countries of Europe, and the United States was less than a week away by boat. In just four months, the “Russian flu” had gone full circle around the world. Little Adolfo was probably exposed when the flu hit Italy in 1890; he was not yet one year old.
“If the hypothesis is correct, Adolfo’s immune system was imprinted by the pandemic of ’89–90,” says Craig. When the 1918 flu, which was presumably a different subtype, came along, his body tried to fight it off, but brought the wrong weapons: “His body was responding, but it was primed to respond to the other flu. It didn’t deal well with the flu that he encountered in 1918.”
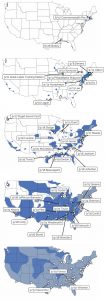
Pandemic’s Progress
Maps prepared by Ruth Craig ’74 and colleagues James Adams and Stephen Gaughan show how the Spanish flu hopscotched through military bases to blanket the country in barely a month, from late August to the end of September.
To test this hypothesis, Alain Gagnon, a professor of demography at the University of Montreal, along with Earn and other colleagues, looked for similar mortality patterns in other flu pandemics. Their results suggest that the 1918 flu was not unique: People born near the time of the 1918 pandemic were likelier to die during a 1957 “Asian flu” pandemic, and people born during that outbreak, in turn, were at greater risk during the 2009 swine flu pandemic.
A study published in Science in 2016 gave new support to the imprinting hypothesis. In that study, researchers looked at flu infection data from China, Egypt, Cambodia, Indonesia, Thailand and Vietnam. They figured out the “first flus” for every birth year between 1918 and 2015 and then compared that data against flu illnesses and deaths for two different flu types. Just as the imprinting hypothesis predicted, people were more likely to get seriously ill or die from flu subtypes that were very different from their first flu.
These correlations are suggestive, but they aren’t conclusive, points out immunologist Matthew Miller, who collaborates with Gagnon and Earn at McMaster. “We see it in epidemiological data, but there’s still not a biological explanation of what’s causing that to happen.” And the epidemiological data still leave key questions open: Perhaps the critical flu exposure happens before a baby is even born, says Miller, when a pregnant woman is infected with flu, sapping her body of resources that would normally be directed to the developing fetus. Or perhaps being exposed to any virulent disease as a baby imparts a lifelong fragility, normally invisible, that makes a person more vulnerable to future illness of any kind. Miller and his colleagues are currently working on testing these ideas.
Yet Miller can sketch out a rough story of what might have happened to Adolfo and other young-adult victims of the 1918 flu, if the “first flu” imprinting hypothesis is correct. “People who were exposed to the 1890 virus would have made antibodies against that virus and T-cells against that virus,” says Miller, describing proteins and immune cells that fight off infections. When the 1918 flu came around, their bodies could have responded with a rush of antibodies and T-cells that “remembered” the earlier flu. But the defensive assault might backfire: The mismatched antibodies would be ineffective, and the T-cells could run riot, making the victim sicker and sicker.
There may be no single explanation for what made young people like Adolfo Sartini so vulnerable in 1918; imprinting probably combined with other factors to create a particularly deadly risk profile. After all, most people who got the 1918 flu, even those born around 1889 and 1890, recovered just fine.
But in Adolfo’s story, Craig sees the shadow of MCL1 and wonders: Did MCL1 help his immune cells “remember” the Russian flu, and did that memory make it harder for him to clear the lung infection that took his life? Did it help unleash a deadly cytokine storm?
To Dockrell, it’s plausible that, at a minimum, Adolfo’s childhood infection with Russian flu may have made him more susceptible to complications of Spanish flu. In fact, in still-unpublished research, Dockrell and his colleagues have found that the flu virus can dial up MCL1 in lung cells, possibly making them more vulnerable to bacterial infections like pneumonia.
“This is total speculation, but in my mind the immune imprinting hypothesis and the cytokine storm hypothesis are not mutually exclusive,” Craig says. “They could both have been at work in 1918.”
Whatever made the 1918 pandemic so deadly, one thing is clear: There will be a next time. And the more researchers know about what happened in 1918, the better prepared we will be to protect ourselves from future pandemics.
AFTER ADOLFO DIED IN VIRGINIA, his body was brought back to Boston, and he was buried at St. Michael Cemetery, a largely Italian cemetery about five miles south of downtown Boston and a few miles west of the Atlantic shoreline. On a chilly day in March, Craig and Bob met there and found Adolfo’s grave, which is packed close with others dated 1918: young children, elderly people, and many in the prime of life, like Adolfo.
A century later, memories of the 1918 flu are mostly like this—gravestones, fading certificates, old Army trunks. But among the headstones at St. Michael, Adolfo’s stands out. It is a granite cylinder carved with winding vines. At the top, it is cut rough. To Craig, it looks like a toppled column: a monument to a life built up and struck down. But the granite is surprisingly smooth, and the flowers she lays are fresh.
The story could begin again tomorrow.